 May, 2 2026
May, 2 2026
Opioid-Induced Pruritus Treatment Efficacy Calculator
Select a treatment option below to analyze its effectiveness in managing Opioid-Induced Pruritus.
Diphenhydramine
Antihistamine (H1 Blocker)Traditional first-line defense, often prescribed but limited by sedation and low efficacy for neural itch.
Naloxone Infusion
Mu AntagonistDirectly blocks receptors. Effective but requires careful titration to avoid reversing pain relief.
Nalbuphine
Mixed Mu/Kappa AgentBlocks itch-triggering mu receptors while activating kappa receptors for mild analgesia.
Butorphanol
Kappa Agonist/Mu Antag.Highly effective in reducing severity scores, particularly noted in cesarean delivery settings.
IV Lignocaine
Sodium Channel BlockerA non-opioid alternative that works via sodium channel blockade. Requires cardiac monitoring.
CR845 (Difelikefalin)
Peripheral Kappa AgonistExperimental Phase II drug. Targets peripheral receptors to minimize central side effects.
Treatment Name
Mechanism
Analysis Results
- Clinical Efficacy Rate --%
- Key Advantage: --
- Clinical Consideration: --
Imagine you just had major surgery. The pain is finally under control thanks to morphine or another opioid. You close your eyes, ready to rest, but then it starts. A crawling, burning sensation on your face and upper chest that makes you want to scratch until the skin breaks. This isn’t an allergy. It’s Opioid-Induced Pruritus, a condition defined as an unpleasant, irritating sensation of the skin that provokes an urge to scratch, occurring as a direct side effect of opioid administration. It affects up to 100% of patients receiving spinal opioids and roughly 30-50% of those getting intravenous doses. For decades, doctors blamed histamine. Today, we know the story is much more complex-and knowing the difference changes how we treat it.
The Great Debate: Is It Just Histamine?
For years, the medical consensus was simple: opioids cause mast cells in your skin to burst, releasing histamine, which causes itching. This theory gained traction after McLelland's 1986 JAMA Dermatology article systematically documented morphine-induced urticaria. Later, researchers like Barke and Hough showed in their 1993 Life Sciences review that drugs like morphine, codeine, and meperidine could trigger this release at specific concentrations.
But here’s the catch: antihistamines don’t work well for most people with this itch. First-generation drugs like diphenhydramine only help 20-30% of patients. Why? Because clinical evidence suggests that for many, especially those receiving standard therapeutic doses, histamine isn’t the main driver. An analysis in QJM (2003) explicitly stated that histamine release from dermal mast cells is not responsible for itch induced by clinical doses of opioids. Instead, the problem lies deeper-in your nerves.
The Neural Pathway: Mu Receptors and TRPV1 Neurons
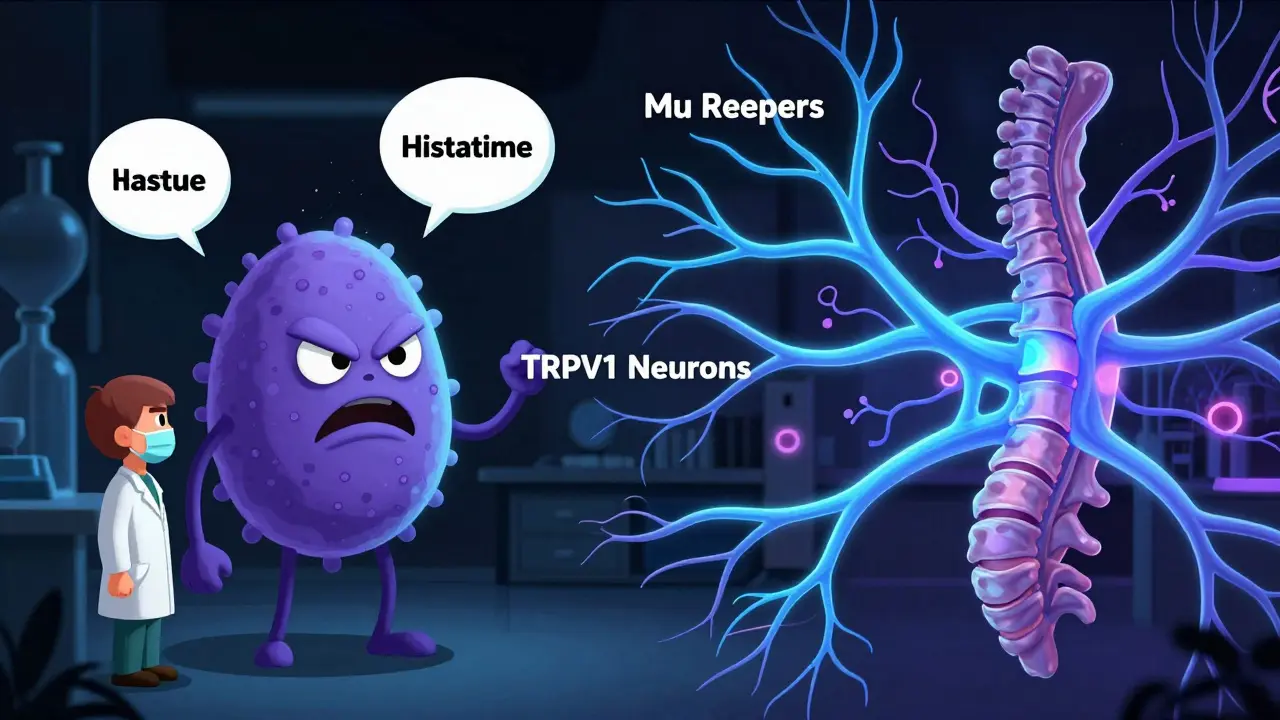
If histamine isn’t always the culprit, what is? Contemporary research points to a dual-pathway mechanism involving the central and peripheral nervous systems. The primary trigger is the activation of the mu opioid receptor (MOR). Unlike pain pathways, which opioids inhibit, itch pathways are activated by these same receptors.
A landmark study published in Nature Scientific Reports (2018) clarified this neural mechanism. Researchers found that mu opioid receptor agonists selectively trigger itch through peripheral nerve fibers emanating from TRPV1-expressing dorsal root ganglion (DRG) neurons. They used DAMGO, a highly selective MOR agonist, to produce quantifiable itch responses. Crucially, this itch was inhibited by naloxone-methiodide, a peripherally restricted opioid receptor antagonist, proving the signal originated outside the brain but was mediated by opioid receptors on nerve endings.
This discovery explains why scratching doesn’t always relieve the itch-it’s not just a surface-level irritation. It also explains why the face and upper torso are the most common sites; these areas have the highest density of MORs. As Dr. Arendt-Nielsen’s team concluded, nerve fibers essential for this response can be eliminated by chronic resiniferatoxin treatment, fundamentally shifting the paradigm away from purely histaminergic explanations.
Why Antihistamines Often Fail
Because the neural pathway plays such a large role, blocking histamine alone is often insufficient. This creates a frustrating cycle for patients. You get prescribed Benadryl (diphenhydramine), feel drowsy for 45 minutes, and still can’t stop scratching. According to a nurse practitioner with 12 years’ experience in post-anesthesia care units, diphenhydramine causes significant sedation without resolving the core issue for most patients.
Second-generation antihistamines like cetirizine are being investigated in prospective double-blind trials (NCT01825655). However, as of 2023, no conclusive pediatric data existed, and their efficacy remains limited compared to receptor-targeted therapies. While they may help patients with comorbid allergies, they are not the gold standard for pure opioid-induced pruritus.
Effective Management: Targeting the Receptor
To truly stop the itch without losing pain relief, you need to target the mu opioid receptor directly. The goal is to block the itch signal while preserving the analgesic (pain-blocking) effect. This requires precise titration because there is a narrow therapeutic window.
| Treatment | Mechanism | Efficacy | Key Considerations |
|---|---|---|---|
| Diphenhydramine | H1 Blocker | 20-30% | Causes significant drowsiness; limited utility |
| Naloxone | Mu Antagonist | 60-80% reduction | Must be dosed carefully (0.25 mcg/kg/min) to avoid reversing pain relief |
| Nalbuphine | Mixed Mu Antagonist/Kappa Agonist | 85% efficacy | Works within 5 minutes; preferred first-line agent in many protocols |
| Butorphanol | Kappa Agonist/Mu Antagonist | High (reduces severity score from 8.2 to 2.1) | Effective in cesarean delivery settings |
| IV Lignocaine | Sodium Channel Blocker | 70% efficacy | Requires cardiac monitoring; non-opioid alternative |
Nalbuphine stands out as a top choice. As a mixed mu antagonist/kappa agonist, it blocks the itch-triggering mu receptors while activating kappa receptors, which can provide mild analgesia themselves. At doses of 5-10 mg IV, it demonstrates 85% efficacy. In contrast, pure antagonists like Naltrexone (1.2 mg IV) achieve 75% symptom reduction but carry a higher risk of compromising pain control if not monitored closely.
Clinical Reality: Timing and Misdiagnosis
Even the best drug fails if given too late. Effective intervention requires administering antipruritic agents within 5-10 minutes of symptom onset. Institutions using standardized protocols, such as the University of Copenhagen's 'Pruritus First Response Algorithm,' report a 40% reduction in rescue medication needs.
A critical danger in managing this condition is misdiagnosis. Thirty-two percent of clinicians initially mistake severe opioid-induced pruritus for anaphylaxis. This leads to unnecessary epinephrine administration, which can cause dangerous spikes in blood pressure and heart rate. Differentiating between the two is vital: opioid-induced pruritus typically presents with facial and upper body itching without respiratory distress, hypotension, or widespread hives (unless histamine release is also occurring).
Patient experiences highlight the severity of this issue. In postpartum forums, 78% of mothers receiving intrathecal morphine described the itching as 'severely disruptive' to early mother-infant bonding. One patient noted, 'I'd rather have pain than feel like fire ants are crawling under my skin 24/7.' This level of distress leads to premature opioid discontinuation in 22% of cases, undermining pain management goals.
Future Directions: Peripheral Kappa Agonists
The field is moving toward more targeted therapies. Current developments include Phase II trials (NCT05217891) of CR845 (difelikefalin), a peripherally restricted kappa opioid agonist. Early results show a 65% reduction in pruritus without central side effects like sedation or respiratory depression. This approach aligns with the European Journal of Pain's 2023 prediction of a paradigm shift toward 'receptor-subtype selective modulation.'
By 2028, experts predict that mu antagonist/kappa agonist combinations will become standard in 75% of major medical centers. Meanwhile, second-generation antihistamines will likely maintain a niche role for the approximately 15% of patients with comorbid allergies, representing a segment of the $480 million annual market for opioid-induced pruritus treatments.
Summary of Key Takeaways
- Opioid-induced pruritus is a common side effect affecting 30-100% of patients, depending on the route of administration.
- While histamine release plays a role in some cases, the primary mechanism involves mu opioid receptor activation on TRPV1-expressing neurons.
- Antihistamines like diphenhydramine are largely ineffective (20-30% success rate) and cause unwanted sedation.
- Nalbuphine and naltrexone are more effective because they directly target opioid receptors, offering 60-85% symptom relief.
- Timing is critical: interventions should begin within 5-10 minutes of symptom onset to maximize efficacy.
- Clinicians must distinguish pruritus from anaphylaxis to avoid inappropriate use of epinephrine.
Why does morphine make me itch if it helps with pain?
Opioids work by binding to mu opioid receptors (MORs). While this binding inhibits pain signals in the spinal cord, it simultaneously activates itch pathways in peripheral nerves, particularly those involving TRPV1-expressing neurons. This paradoxical effect means the same mechanism that relieves pain can trigger intense itching.
Is opioid-induced itching an allergic reaction?
No, it is not typically an allergic reaction. While some opioids can cause histamine release leading to hives, true opioid-induced pruritus is primarily a neural phenomenon driven by receptor activation. Allergic reactions usually involve respiratory distress, swelling, or hypotension, whereas pruritus is localized to the skin, often on the face and upper body.
What is the most effective medication for stopping opioid itch?
Nalbuphine is considered one of the most effective treatments, with an 85% efficacy rate. It works as a mixed mu antagonist/kappa agonist, blocking the itch signal while providing mild pain relief. Naltrexone and low-dose naloxone infusions are also highly effective but require careful monitoring to ensure pain control is not compromised.
Do antihistamines like Benadryl work for this type of itching?
Generally, no. First-generation antihistamines like diphenhydramine are only effective in 20-30% of cases because the itch is not primarily caused by histamine. Additionally, they cause significant drowsiness, which can interfere with recovery and monitoring.
How quickly do treatments for opioid-induced pruritus start working?
For optimal results, antipruritic agents should be administered within 5-10 minutes of symptom onset. Medications like nalbuphine can work within 5 minutes when given intravenously. Delaying treatment reduces efficacy and increases patient discomfort.
